Overview
Senior Investigator
Research Areas (IRP Lab Groups)
Vasculitis is a family of rare diseases defined by inflammation in blood vessels. These conditions, if not quickly recognized and properly treated, can cause serious and life-threatening damage to multiple organ systems. The goal of the Vasculitis Translational Research Program (VTRP) is to discover factors that cause vasculitis and to develop novel ways for physicians to diagnose and monitor these diseases more effectively.
Tremendous progress over the last few decades has transformed vasculitis from frequently fatal into a chronic, manageable illness. However, treatment with potentially toxic medications, including steroids and other medications that suppress the immune system, is generally required to induce remission. Although most patients with vasculitis achieve remission with treatment, the majority of patients experience one or more recurrences of the disease. Disease relapse can result in additional permanent organ damage and can be fatal.
At present, we have a very crude understanding of what causes vasculitis. There are very few tests available that reliably help a doctor predict which patients will experience a relapsing disease course. The identification of factors that cause vasculitis and the discovery of biomarkers that can predict clinical outcomes and guide patient-specific therapeutic decisions is the major focus of the VTRP.
The VTRP was established in 2013 to evaluate patients with known vasculitis or with clinical suspicion of vasculitis. Diseases of interest include Takayasu’s arteritis (TAK), relapsing polychondritis (RP), giant cell arteritis (GCA), granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), eosinophilic granulomatosis with polyangiitis (EGPA), and polyarteritis nodosa (PAN) among other types of vasculitis.
Current Projects
A selection of our current projects are as follows:
VEXAS Syndrome
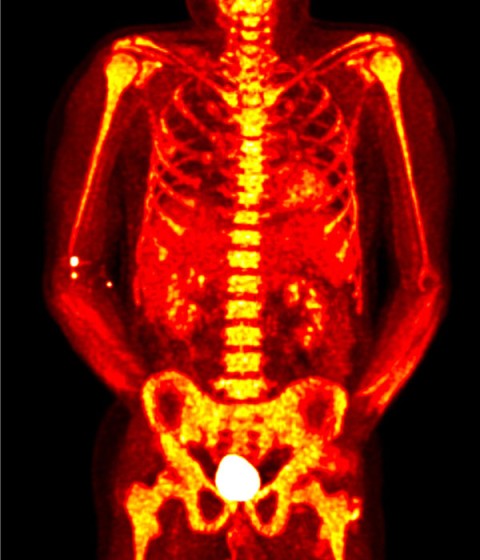
VEXAS syndrome is a disease with very heterogeneous clinical phenotypes, including inflammatory and hematologic manifestations. It is caused by mutations in the ubiquitin-activating enzyme E1 (UBA1) gene of blood cells. VEXAS patients can present with different UBA1 mutation profiles, including three canonical mutations at Met41. We have observed that these mutation types are associated with specific presentations of the disease.
Using genomic techniques, we aim to increase our understanding of the pathogenesis of VEXAS syndrome. Through ongoing analyses and future research, we are working to uncover the impact of the UBA1 mutation on hematopoietic cells. We hope that this understanding will lead to the development of more targeted treatments. In parallel, we are working to describe and classify the clinical heterogeneity and natural history of VEXAS syndrome.
Find out how this syndrome is diagnosed, who gets it, and learn about the ongoing work being done to understand and treat it – including the NIAMS clinical VEXAS cohort and the work being done by the VTRP, and other additional resources.
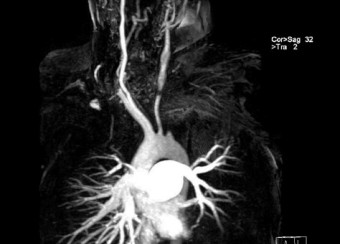
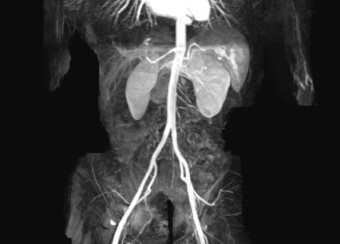
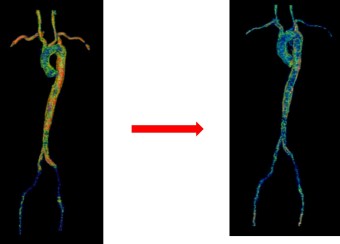
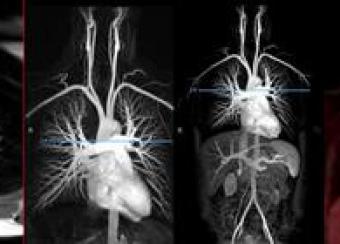
Vascular Imaging in Large-Vessel Vasculitis

Takayasu’s arteritis and giant cell arteritis are the two major diseases that affect the aorta and its primary branches. The detection of active vasculitis is important to prevent the progression of vascular disease and organ damage; however, clinical assessment of disease activity is challenging. Although vascular imaging is considered essential in these diseases, there are no accepted guidelines for imaging and prospective imaging data is lacking.
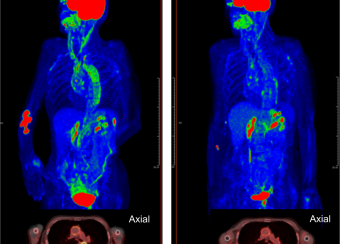
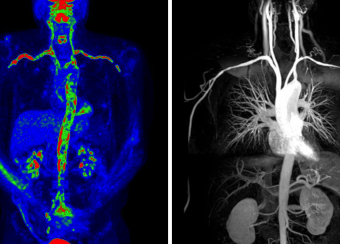
Our group has employed advanced molecular imaging techniques in patients with large-vessel vasculitis to study whether serial imaging has diagnostic and prognostic value in addition to monitoring utility. We discovered that the majority of patients with large-vessel vasculitis have evidence of ongoing vascular inflammation by FDG-PET scans during periods of apparent clinical remission and that the degree of vascular inflammation seen during clinical remission predicts future relapse.
We developed and validated PETVAS, a quantitative measure of vascular inflammation that is being used internationally as an outcome measure in ongoing clinical trials. Our lab is working to define a potential immunologic basis for subclinical inflammation in these conditions.
Relapsing Polychondritis

Recently, our group established the largest prospective cohort of relapsing polychondritis (RP). The NIH RP program currently has recruited >100 patients with this disease. Patients are evaluated at 6-month intervals using a standardized assessment protocol with biobanking of research samples. From these efforts, we have published some of the only observational cohort data for RP.
Using latent class analysis, we have defined three major clinical subsets of RP based on the pattern of organ involvement and we are using these subsets as the framework to investigate causal disease factors, treatment approaches, and potential biomarkers. Recently, in collaboration with the Kastner lab, we discovered a genetic factor that is causal in 8% of patients with relapsing polychondritis.
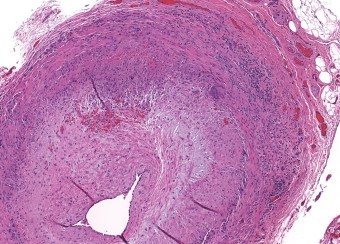
Neutrophil Biology in Small and Medium-Vessel Vasculitis

Neutrophils are key cells that drive inflammation in many forms of vasculitis. Our group has partnered with the Kaplan Lab in the Systemic Autoimmunity Branch to study novel aspects of neutrophil biology and innate immune system involvement in ANCA-associated vasculitis, drug-induced vasculitis, and monogenic vasculitis.
Through these efforts, we have learned that a subset of neutrophils known as low-density granulocytes may play a pathogenic role in ANCA-associated vasculitis, that cholinergic receptors on neutrophils mediate disease in levamisole-induced vasculitis, and that the adenosine-mediated activation of neutrophils contributes to inflammation in deficiency in deaminase 2 (DADA2).
Scientific Advances
Core Research Facilities
Labs at the NIAMS are supported by the following state-of-the-art facilities and services:
Staff
Image & Media Gallery
Clinical Trials
Vasculitis is a group of diseases that inflame and damage blood vessels and tissue. It can cause many medical problems. Few tests can diagnose the disease, and none can reliably predict a relapse. Researchers want to study people s genes and follow people over time to see how the disease affects them.
Scientific Publications
Selected Recent Publications
Key Publications
News & Highlights
Skin Features Provide an Early Clue to the Diagnosis of VEXAS Syndrome
Scientists at NIAMS and other institutions have shed light on patterns of skin characteristics seen in patients with