Overview
Earl Stadtman Investigator
Research Areas (IRP Lab Groups)
The Functional Immunogenomics Section (FIS) studies how genes control the function of cells in the human immune system. We are especially interested in how treatments that change the expression of human genes affect the behavior of immune cells. Our long-term goal is to enable the development of better treatments for human diseases that are caused by an overactive immune system.
FIS scientists work at the intersection of clinical medicine, human immunology, genomics, and bioinformatics. A major area of focus for our team is the dissection of the mechanisms by which glucocorticoids regulate human immunity. Glucocorticoids are a class of drugs. They have been the cornerstone of anti-inflammatory and immunosuppressive therapies for over 70 years. They are highly effective at controlling an overactive immune system, but they also have a long list of serious side effects. Although they are one of the most widely prescribed classes of drugs worldwide, there are surprisingly large gaps in our understanding of how glucocorticoids regulate the behavior of human immune cells. Our team is working to help fill in those gaps, by applying advanced genomic and proteomic technologies to the study of the response to glucocorticoids in healthy humans and people with autoimmune diseases.
Our Research Platform

The FIS’s research platform combines clinical studies in patients with autoimmune diseases and healthy volunteers, with laboratory studies of human cells and tissues. Cutting-edge genomic and proteomic technologies are employed to identify the key genes and proteins that are responsible for the functional effects of glucocorticoids in human cells. In collaboration with the National Center for Advancing Translational Sciences (NCATS), we then work to translate the findings of our research into therapies that can mimic the clinically beneficial effects of glucocorticoids but have fewer side effects.
Bioinformatics Tools
Web applications developed by our lab:
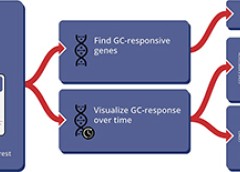
GCgx - How do glucocorticoids work?
GCgx is a scientific web application that allows scientists to study the response of different cell types to glucocorticoids. It provides a simple, mobile-friendly tool that can offer quick answers to the following questions: Is my gene of interest responsive to glucocorticoids in specific cell types? If so, how does its level of expression vary over time and how statistically significant is the change?
NeutGX - Explore the neutrophil transcriptome
NeutGX is a web application that allows investigators to explore the transcriptome of human neutrophils. This tool is available as part of a suite of NIAID Bioinformatics Applications.
Core Research Facilities
Labs at the NIAMS are supported by the following state-of-the-art facilities and services:
Staff

Our lab is always seeking to recruit the best talent in the scientific community. If you are interested in joining our team, please contact us.
Former Lab Members
Postbaccalaureate Students
- Rose Dever, B.S., 2023-2025
- Javid Javadikhomami, B.S., 2023-2025
- Robert Kwiat, B.S., 2021-2023
- Qilin Cao, B.S., 2019-2022
- Thai Tran, B.S., 2019-2021
Medical Research Scholars
- Ariel Bohner, D.V.M., 2021-2024
Rotating Medical/Graduate Students
- John Michael Sanchez, M.D. Ph.D. Student, 2021