Overview
Emeritus Investigator
Research Areas (IRP Lab Groups)
While this laboratory has previously focused on characterizing the transcriptional regulatory networks and signaling pathways linked to epidermal differentiation and wound healing, Dr. Morasso is no longer conducting active research and is now an Emeritus Investigator.
Epidermal Differentiation and Inflammatory Disorders
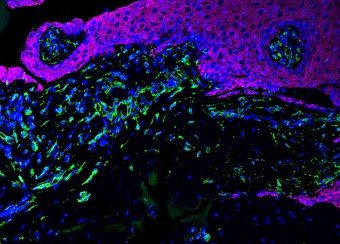
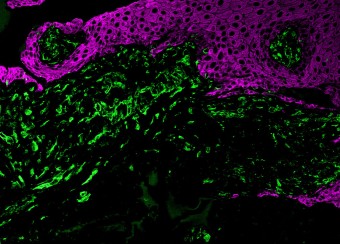
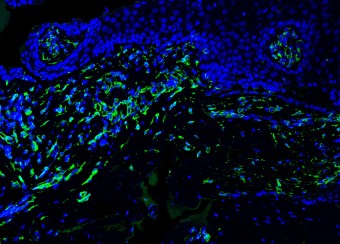
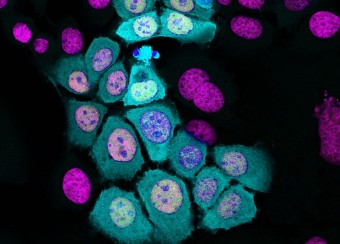
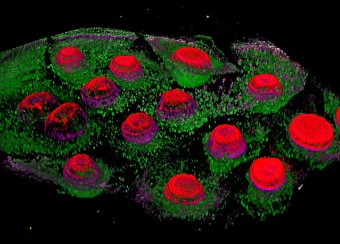
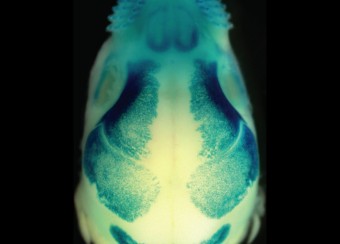
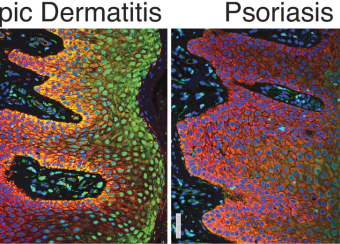
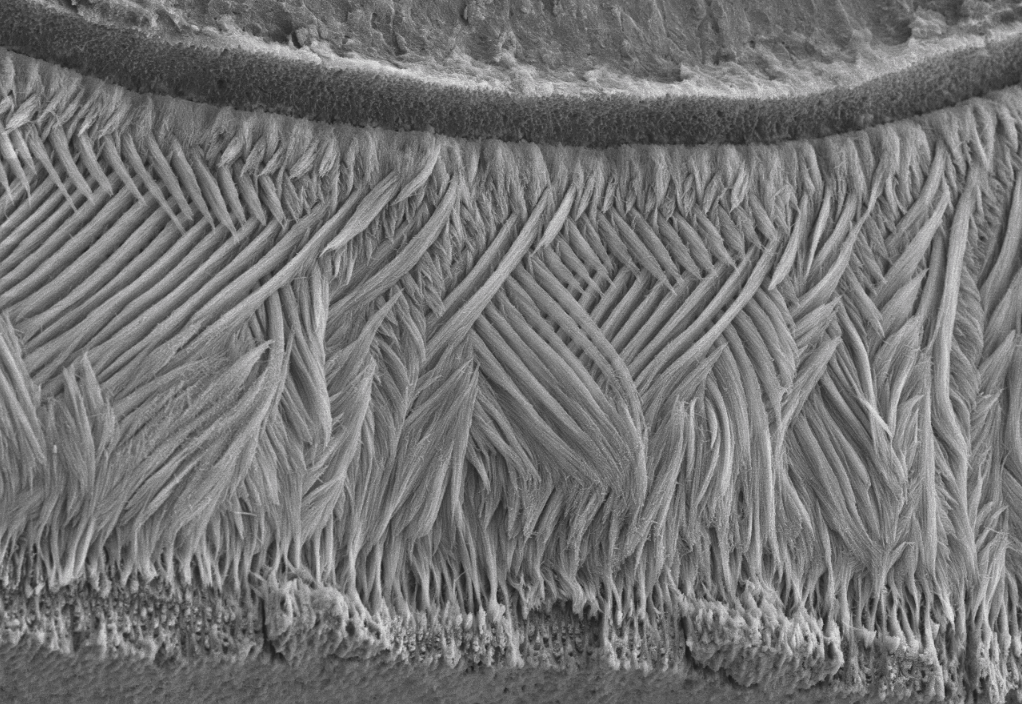
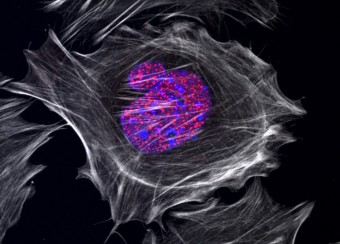
Epidermal homeostasis results from a balance between keratinocyte proliferation and differentiation, with alterations to this balance leading to cancer. Using animal models, we have demonstrated the central role of the transcriptional regulator during epidermal differentiation. Through a combination of transcriptomic and bioinformatic analyses, we have identified a DLX3-dependent network that regulates cell cycle and the activated ERK- and STAT3-dependent signaling pathways that are crucial to maintaining cutaneous homeostasis. The epidermal conditional deletion of DLX3 leads to the disruption of skin barrier formation. It is linked to epidermal hyperplasia and the development of an inflammatory response, characterized by the accumulation of IL-17-producing T cells. Our studies also provide a novel understanding of the signaling networks regulating squamous tumorigenesis.
Wound Healing

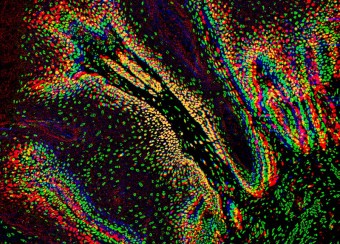
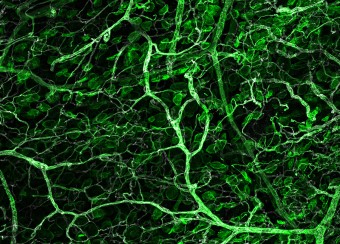
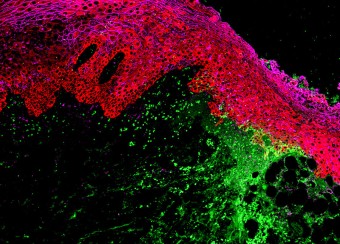
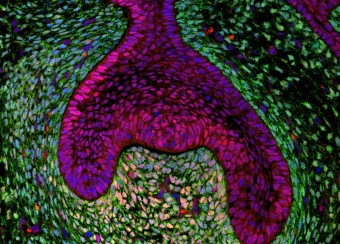
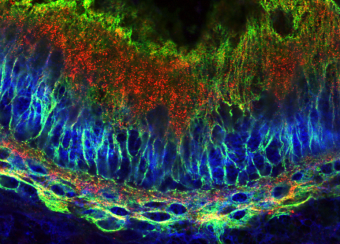
Oral wounds are considered an ideal prototype of adult tissue repair due to their intrinsic ability for scarless accelerated wound healing. Our studies have shown that wound-activated transcriptional networks are active in oral mucosa at a basal state, priming this tissue for wound repair by controlling epithelial cell differentiation. We have determined the capability of the SOX2 and PITX1 transcription factors to reprogram cutaneous keratinocytes for accelerated wound resolution. Furthermore, we found that SOX2 promotes keratinocyte migration and angiogenesis through the upregulation of EGFR ligands and activation of the ERK/MAPK pathway. In contrast to acute cutaneous and oral wounds, chronic non-healing wounds do not resolve and are characterized by deregulated inflammation. This is due to ineffective recruitment and activation of macrophages and neutrophils. We are currently focused on identifying pathways responsible for the downregulation of inflammation that will be pivotal for diagnostics and the development of potential therapeutics.
Ectodermal Dysplasias and Ectodermal Appendage Development
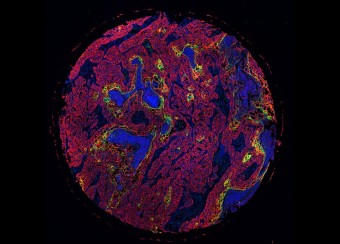
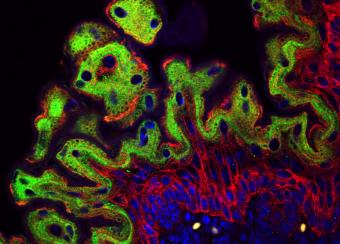
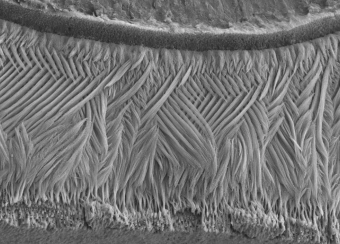
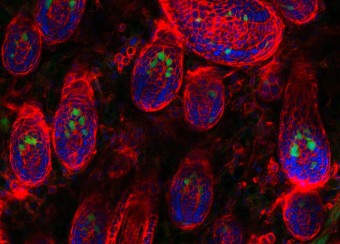
Ectodermal dysplasias are a group of heritable pathological disorders that result from anomalies in epithelial/mesenchymal-derived appendage formation. The importance of DLX3 is underscored by the effects of mutations found in patients with autosomal dominant Tricho-Dento-Osseous (TDO) syndrome. We have demonstrated the crucial role of DLX3 in epidermal development and tooth and hair formation through direct regulation of specific sets of keratins. Using genetic and intraoral examination data from human patients, we identified several missense polymorphisms in keratins that lead to a higher risk for dental caries.
Scientific Advances
Core Research Facilities
Labs at the NIAMS are supported by the following state-of-the-art facilities and services:
Staff
Former Lab Members
Postdoctoral Fellows
- Daniela Grassini, Visiting Fellow (2018-2021)
- Akihiko Uchiyama, Visiting Fellow, JSPS Fellowship (2016-2019)
- Shreya Bhattacharya, Visiting Fellow (2014-2017)
- Kathleen Baysac, IRTA Fellow (2015-2017)
- Ramiro Iglesias-Bartolome, Research Fellow (2015-2016)
- Paul Bible, IRTA Fellow (2013-2015)
- Jin-Chul Kim, Visiting Fellow (2010-2015)
- Juliane Lessard, IRTA Fellow (2013-2014)
- Elisabetta Palazzo, Visiting Fellow (2011-2014)
- Juliane Isaac, Visiting Fellow (2009-2013)
- Junko Okano, Visiting Fellow (2008-2012)
- Olivier Duverger, Visiting Fellow (2006-2011)
- Joonsung Hwang, Visiting Fellow (2004-2011)
- Oleksandr Kalinin, Visiting Fellow (2004-2008)
- Valentina Pietroni, Visiting Fellow (2006-2008)
- Nadezda Radoja, IRTA Fellow (2004-2006)
- Meeyul Hwang, Visiting Fellow (2001-2005)
- Janine Bryan, IRTA Fellow (2000-2002)
- Geon Tae Park, Visiting Fellow (2000-2003)
Postbaccalaureate Fellows
- Emma Hope (2020-2022)
- Stephen Worrell (2020-2021)
- Michael Cross (2017-2019)
- Rose Graf (2017-2018)
- Veronica Nagle (2016-2017)
- Maya Evanitsky (2016-2017)
- Chelsea Karacz (2015-2017)
- Meghan Kellett (2013-2016)
- Victoria Sun (2014-2015)
- Takahiro O'Hara (2012-2014)
- Anna Gormley (2012-2013)
- Claire Levy (2011-2012)
- Angela Zah (2010-2012)
- Anne Bartels (2009-2010)
- Ryosuke Kita (2009-2010)
- Nicole Gentile (2007-2008)
- Susie Chen (2007-2008)
- Delia Lee (2006-2007)
- Sarah-Jo Stimpson (2005-2006)
- Charlene Rivera (2001-2002)
- Taraneh Mehrani (2000-2001)
Research Fellows
- Subhashree Nayak, Research Fellow (2017-2024)
- Deepti Bajpai, Research Fellow (2017-2022)
Staff Scientist
- Olivier Duverger, Staff Scientist (2011-2017)