Overview
Clinical Director
Research Areas (IRP Lab Groups)
The long-term goals of the Pediatric Translational Research laboratory are to understand cellular and molecular mechanisms underlying the pathogenesis of spondyloarthritis. Spondyloarthritis refers to a spectrum of chronic immune-mediated inflammatory diseases that affect an estimated 1% of the population. They differ from other types of inflammatory arthritis in genetic predisposition, pathogenesis, and long-term outcome. Spondyloarthritis often begins as an undifferentiated disease that, in many cases, progresses to ankylosing spondylitis (AS). AS can lead to ankylosis of the spine and significant disability. Although most people affected by AS are adults, approximately 15-20% are first affected as children. This juvenile onset AS (JAS) differs from adult onset disease because spinal (axial) symptoms are usually missing, while peripheral arthritis and enthesitis are more prominent. Most spondyloarthritis in children is classified as a form of juvenile idiopathic arthritis known as enthesitis-related arthritis (ERA) or psoriatic arthritis (PsA). However, children with ERA or PsA and spinal involvement are not distinguished from those with only peripheral arthritis and enthesitis. Since individuals with axial involvement are most likely to progress to AS, recognizing early spinal involvement in children is paramount.
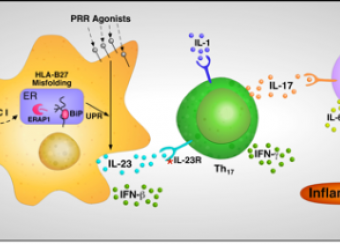
The development of AS is primarily determined by one’s genetic make-up. HLA-B (predominantly the B27 allele) plays a major role, accounting for an estimated 30-40% of overall susceptibility, with additional genes, such as ERAP1, IL23R, IL1RII, IL1 (probably IL1A), CARD9, TNFR1, TRADD, STAT3, PTGER2, ANTXR2 and others also contributing. Our knowledge of how these genes and their variants work together to promote the inflammation and bone formation that occurs in AS is incomplete, although new targets have been identified. As therapeutic options improve, identification of children with spondyloarthritis who have axial involvement will be increasingly important, as they may benefit the most from early aggressive intervention.
Current projects include:
- Evaluating the role of HLA-B27 and other genetic factors in disease.
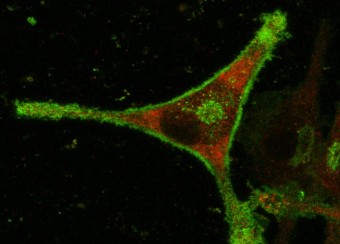
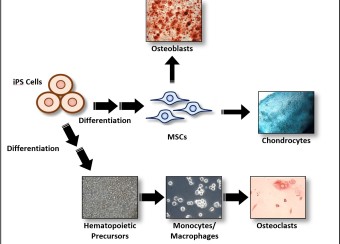
- Studies employ a transgenic animal model of spondyloarthritis and cells from human subjects including induced pluripotent stem cells (iPSC), to evaluate effects of HLA-B27 expression and misfolding on ER stress and its consequences for cytokine production and cellular function.
- Effects on osteoclast and osteoblast development and function.
- Elucidating genes that influence how HLA-B27 causes spondyloarthritis in rats.
- Determining how ERAP1 affects spondyloarthritis pathogenesis.
- Understanding how HLA-B27 shapes the immunology and microbiology of the gut.
- Determining clinical manifestations and biomarkers of early axial spondyloarthritis in children.
These studies will inform us on mechanisms that drive pathogenesis of spondyloarthritis and suggest novel strategies that can be developed to more effectively treat disease.
Core Research Facilities
Labs at the NIAMS are supported by the following state-of-the-art facilities and services:
Staff
Image & Media Gallery
Clinical Trials
Researchers want to see if people with RA in remission on a TNF inhibitor can stay in remission without this medicine. Also there may be a clinical, imaging (MRI, ultrasound), laboratory profile that will help to determine which patients remain in remission after stopping these drugs. This study will provide important new information on the best treatment approach for patients with RA in remission.