This document outlines the process by which requests for access to existing biospecimens from the OAI resource are processed, evaluated, and approved.
Types of Specimen Available
| Type of Specimen | Maximum Allowable Specimen Per Study |
|---|---|
| Serum | 0.15 ml (total of serum and plasma per study) |
| EDTA plasma | 0.15 ml (total of serum and plasma per study) |
| Citrate plasma | 0.15 ml (total of serum and plasma per study) |
| DNA | 500 ng |
| RNA | To Be Determined |
| Urine | 2 ml |
| Pax-Gene tubes for RNA extraction | |
| Whole blood is not available. |
Review and Approval Process
Individuals who have agreed to serve on the Biospecimen Review and Allocation Committee (BRAC) will be asked to make recommendations to NIAMS regarding approval of proposals that request OAI biospecimens. BRAC review panels will consist of 2-3 voting members appointed by the NIAMS Project Office (PO).
Proposals will be reviewed for:
- Feasibility (i.e., availability of the type and amount of specimen(s) requested)
- Scientific merit based on modified NIH criteria
- Consistency with the objectives and goals of the OAI.
- Priority for proposals requesting serum or plasma will be given to those proposing longitudinal measurements of biomarkers.
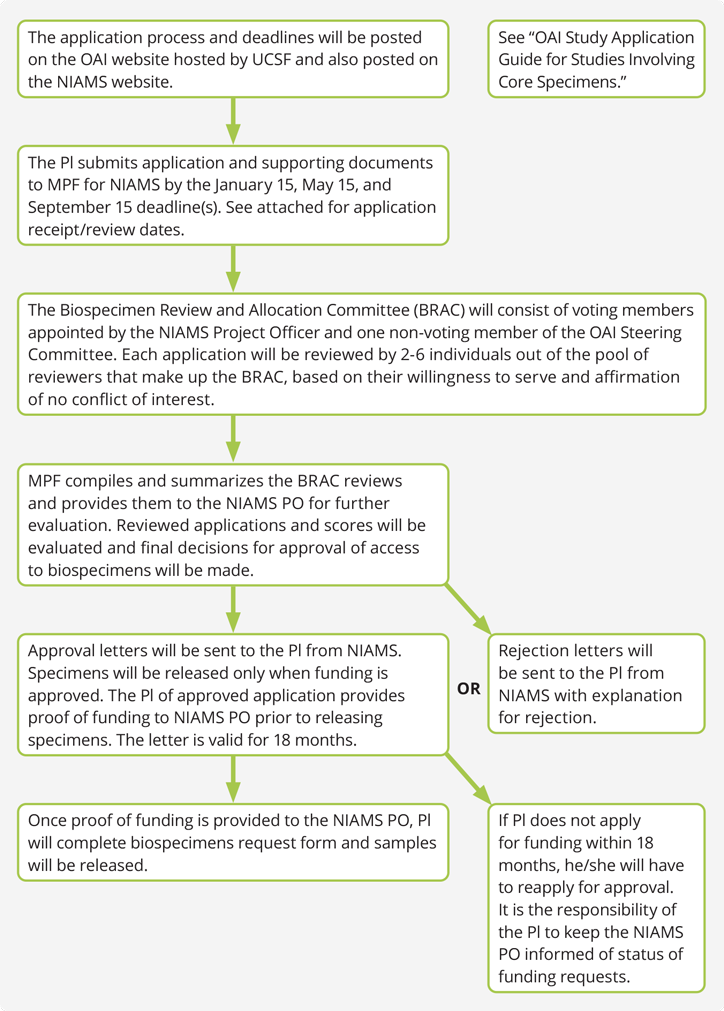
Applicants will be informed of approval or disapproval by the BRAC. Applications that are not approved may be resubmitted with additional data or modifications as recommended. The NIAMS PO will issue a letter of approval or disapproval after reviews are completed. The release of the biospecimens will be contingent on verification of funding for the proposed project. A diagram of the application and review process is shown below. The PI will be expected to provide sufficient funds to cover costs of sample pulling and shipping when such services are rendered.
How to Apply
- Complete the Osteoarthritis Initiative Biospecimen Request Form.
- Submit the request form, narrative proposal (link to section below), list of requested OAI sample ID’s or ID’s of matched pairs each with 1--2 alternative samples in case of insufficient volume, and any required attachments to MPF, the administrator for the BRAC, by email to [email protected]. Electronic versions of documents are the preferred method of submission.
- The full review process will take 6-10 weeks. The principal investigator (PI) may receive questions from the BRAC through MPF to clarify the proposal further. Once the BRAC reviews the proposal, it will be forwarded to the NIAMS PO for review and possible approval. The NIAMS PO will provide the final notification of approval to the PI through MPF. If the proposal is not accepted, the PO will provide a written explanation for declining the request. If these concerned can be effectively addressed, a PI may revise and resubmit the proposal.
Once the request is approved, requested specimens will be set aside but will not be dispensed until the NIAMS PO receives a letter from the funding agency (sponsor) confirming that the study will be funded. The PI will receive samples in a blinded manner. The investigator must submit the data to the NIAMS PO for un-blinding. However, these data will not be released to the OAI website until at least one year after the originating investigator has received the un-blinding documentation with OAI participant identifications attached from the NIAMS PO. Access to biospecimens will only be provided with the understanding that data generated through the study proposal will be submitted to the OAI public web site within a reasonable timeline, not to exceed one year post un-blinding.
The PI is responsible for the costs of pulling, aliquoting, and shipping of the samples, and any costs associated with follow up inventory. A maximum of 18 months may lapse between protocol approval and confirmation of funding. If funding confirmation or a status update concerning funding is not provided to the NIAMS PO within 18 months, the hold will be dropped and the samples will be made available to other requests.
Narrative Proposal Format
The narrative proposal format should conform to the following specifications:
- Maximum of 10 pages (including abstract, tables and figures); references may be additional. The PI should to be as concise as possible. Proposals that are longer than 10 pages will be returned for editing.
- Minimum font size of 11 pt (12 pt is preferred).
- Express amount of specimen requested as follows ( the PI must justify sample size and amount of specimen requested; see limitations above):
- Serum, EDTA and citrate plasma in milliliters (ml)
- DNA in nanograms (ng)
- Urine in ml
- RNA in micrograms or nanograms (not currently extracted)
- List genes to be studied by gene name followed by HUGO Gene Nomenclature Committee gene symbol in parenthesis — e.g. low density lipoprotein receptor (LDLR), estrogen receptor 1 (ESR1).
To assure an efficient review process, these points should be clearly and fully described in your submission (while staying within the 10-page limit):
- A 200-word abstract to communicate the key objectives
- Specific aims
- Background and significance, including the potential to contribute to the health of OA patients
- Methods, including all of the following that are applicable:
- Sample type (citrated plasma, EDTA plasma, serum, urine, DNA, RNA), volume, and study years for which samples are being requested (refer to OAI Biospecimen Request Form)
- Requirements for frozen versus previously thawed samples and, if the latter, any limitations on number of freeze thaw cycles
- Description of study participants from whom samples are being requested (number and characteristics))
- Statistical design, analyses and power calculations
- Assays planned (be specific; for SNA, attach list of genes and SNPs following nomenclature described above)
- Methodology description (include references or a detailed description if not yet published) and rationale for the selection of the biomarker assays
- Specificity, sensitivity, and precision for each proposed assay
- Projected timeline for a) pulling & shipping; b)sample analysis; c) return of sample, if applicable
- How will the study be funded? If applying for a grant, who is the sponsor and what is the time line for funding?
- Investigator qualifications and involvement in OA research
- Environment (expertise and capabilities to be able to conduct proposed research)
- Do you anticipate using the data to file an application for a patent, to the FDA, or for other similar purposes?
- Is DNA to be used? If so, include a plan for addressing any relevant clinical, ethical, legal, or social implications of the findings.
- Data sharing plan (Data generated from use of the biospecimens must be returned to the NIAMS PO within one year of un-blinding for posting on the OAI Online website.)
Application Submission Deadlines
Review Criteria
Scoring note: 5 = outstanding, 4 = excellent, 3 = good, 2 = acceptable, 1 = poor, 0 = unacceptable
Scientific Review (Scored 0-5)
Significance: Does this study address an important problem? If the aims of the application are achieved, how will scientific knowledge or clinical practice be advanced? What will be the effect of these studies on the concepts, methods, technologies, treatments, services, or preventative interventions that drive this field?
Approach: Are the conceptual or clinical framework, design, methods, and analyses adequately developed, well integrated, well-reasoned, and appropriate to the aims of the project? Does the applicant acknowledge potential problem areas and consider alternative tactics?
Innovation: Is the project original and innovative? For example: Does the project challenge existing paradigms or clinical practice, or address an innovative hypothesis or critical barrier to progress in the field? Does the project develop or employ novel concepts, approaches, methodologies, tools, or technologies for this area?
Investigator: Are the investigators appropriately trained and well suited to carry out this work? Is the proposed work appropriate to the experience level of the principal investigator and other researchers? Does the investigative team bring complementary and integrated expertise to the project (if applicable)?
Environment: Does the scientific environment in which the work will be done contribute to the probability of success? Do the proposed studies benefit from unique features of the scientific environment, or subject populations, or employ useful collaborative arrangements? Is there evidence of institutional support?
OAI Priorities and Policy (scored 0-5)
- Potential for contributing to the health of osteoarthritis patients or understanding of disease.
- Draws on unique characteristics of the OAI.
- Efficient use of biologic specimens (volume of specimen; number of genotypes/ phenotypes tested; use of high throughput facilities; need for ad hoc thawing).
- Complements the current portfolio of existing studies.
- Value of scientific resources that will be contributed to the OAI.
| III. Operational Criteria (not scored) | Meets Criterion? |
|---|---|
| If necessary, has the proposed use of biologic specimens been submitted for IRB approval? | Y / N / NA |
| Is notification of study results required? If so, is there an appropriate plan to notify participants? | Y / N / NA |
| Is there an appropriate plan for disposition of unused samples? | Y / N / NA |
| Is there an appropriate plan for disposition of study data (e.g., publication, use in larger grant application, etc.) and is there a time line for submission of data to NIAMS for adding to OAI database? | Y / N / NA |
Application and Approval Process for Studies Involving OAI Core Biospecimens
Please note — the stages outlined below assume approval at every review stage. However, at any stage in this review process, the application may be sent back to the PI for revisions or more information may be requested.