
National Institute of Arthritis and Musculoskeletal and Skin Diseases Congressional Justification FY 2025
Department of Health and Human Services National Institutes of Health
An image from NIAMS-funded researchers of a specific type of immune cells, called T peripheral helper (Tph) cells (stained blue). These cells stimulate other immune cells called B cells (green) to produce autoantibodies in the joints of people with rheumatoid arthritis.
Credit: : Drs. M Brenner, D. Rao, at Brigham and Women’s Hospital, Boston, MA
General Notes
- FY 2024 funding levels cited in this document are based on the Continuing Resolution in effect at the time of budget preparation (Public Law 118-35) and do not include HIV/AIDS transfers.
- Detail in this document may not sum to the subtotals and totals due to rounding.
Sections
Director’s Overview

Lindsey A. Criswell,
M.D., M.P.H., D.Sc.
The National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) is the primary Institute supporting biomedical research on diseases of the bones, joints, muscles, and skin. The NIAMS mission is to support research into the causes, treatment, and prevention of arthritis and musculoskeletal and skin diseases; the training of basic and clinical scientists to carry out this research; and the dissemination of information on research progress in these diseases.
Burden of Disease in NIAMS Mission Areas
The diseases and conditions within NIAMS’ mission areas have substantial physical and psychosocial consequences and can have a profound detrimental effect on a person’s quality of life, productivity, finances, and overall health. Moreover, they affect a significant portion of the U.S. population. For example, arthritis, which encompasses a variety of conditions like osteoarthritis, rheumatoid arthritis, spondyloarthritis, and juvenile arthritis, is one of the leading causes of disability in the United States. It affects millions of people, causing joint pain, inflammation, and reduced mobility. Musculoskeletal disorders encompass a wide range of conditions, including osteoporosis, muscular dystrophy, and back pain. These conditions collectively result in tremendous suffering, affecting patients of all ages. Osteoporosis can lead to fractures, joint replacements, and mobility issues, while spondyloarthritis can result in spinal fusion, particularly in our aging population. Similarly, skin diseases, including common conditions such as psoriasis and eczema, can be chronic and debilitating, affecting not only the skin but also a person’s overall well-being. NIAMS conducts research to better understand underlying mechanisms behind numerous diseases in our mission areas and helps to develop innovative treatments.

Credit: iStock
We recognize that many of the diseases within NIAMS’ purview affect women and underserved populations disproportionately. Health disparities unfortunately impact groups that have historically faced, and continue to experience, increased barriers to, or exclusion from, many aspects of life due to race, age, sex, socio-economic status, geographic location, or other factors. We strive to promote health equity by closing the gap of health disparities in our mission areas. A recent study examined the causes of mortality in the United States from 2000 to 2019, with a particular focus on racial, ethnic, and county-level variations. It found that mortality in patients with skin and subcutaneous diseases and musculoskeletal disorders was higher in American Indian or Alaska Native and Black populations compared to White populations.[1]

Credit: Martyn Green, NIAMS
This level of detail offers an opportunity for NIAMS and the research community to focus on where these differences are the most pressing and provides a clearer view towards potential solutions for disparities within our mission areas. In accordance with our focus on eliminating health disparities, NIAMS collaborated with the National Heart, Lung, and Blood Institute to directly address health disparities and better engage underserved populations in research within the local community in Washington, D.C. This partnership is conducted through the existing NIAMS Community Health Clinic. It is particularly important because it allows investigators to directly engage with the community, which will broaden public participation and provide for greater community engagement for this important collaboration. At the new clinic, called the Hope Center as described in a program portrait, clinician-scientists address rheumatologic diseases in diverse populations.
Yesterday, Today, and Tomorrow
Progress in Rheumatic Diseases
Rheumatic diseases affect the joints and are very common, severe, and painful. Historically, treatments for these diseases have focused on decreasing inflammation and slowing down tissue damage, but most patients respond only partially to available therapies. Rheumatic diseases are often complex and variable, making prevention, diagnosis, and treatment difficult.

Credit: iStock
In 2014, NIAMS partnered with the National Institute of Allergy and Infectious Diseases, industry, and non-profit organizations to launch the Accelerating Medicines Partnership® Rheumatoid Arthritis and Systemic Lupus Erythematosus (AMP® RA/SLE) program to discover new biologic targets for therapeutics. This program took a molecular and cellular approach to understanding the underlying biological pathways in two specific rheumatic diseases, RA and SLE. Research supported by this program generated important new knowledge about the mechanisms of these diseases. For example, AMP RA/SLE researchers identified a new core population of CD 8 T immune cells that drive inflammation in RA. The role of these cells was previously unknown, and this study marks the first report of their involvement in inflammatory activity in RA. These findings provide insight into RA and other inflammatory diseases as well as potentially cancer and chronic infection.
Building upon the success of AMP RA/SLE, NIAMS now leads the AMP Autoimmune and Immune-Mediated Diseases (AMP® AIM) program. AMP AIM was created to discover shared inflammatory pathways in RA, SLE, psoriasis, psoriatic arthritis, and Sjögren’s disease, which will advance the search for new treatments. Within the AMP AIM program, NIAMS and the NIH Office of Research on Women's Health, in partnership with the NIH Office of Data Science Strategy, created the Team Science Leadership Scholars Program (LSP). In FY 2023, the LSP announced its inaugural cohort of six scholars whose research focuses on women’s health studies in autoimmune diseases. Through the LSP, the scholars work closely with AMP AIM investigators; leverage the rich infrastructure, samples, and data generated by AMP AIM; and develop strong skills to lead complex, high-level research.
NIAMS also continues to support other research in rheumatic and autoimmune diseases that has significantly advanced the field. For example, NIAMS-funded researchers developed a novel assay that detects a marker for lupus nephritis in urine. Assay results can be read on a smartphone, allowing for an easy way to monitor disease activity and response to therapies. Lupus nephritis is the leading cause of morbidity and mortality in people with SLE; usually, diagnosis requires an invasive kidney biopsy that can lead to complications, so this new technology has the potential to save lives.
Looking forward, NIAMS is partnering with the NIH Office of Autoimmune Disease Research (OADR) to better understand the impact of environmental exposures in autoimmune diseases. NIAMS collaborated with the OADR to issue a Notice of Special Interest for EXposome in Autoimmune Disease Collaborating Teams PLANning Awards (EXACT-PLAN). The program’s goal is to develop a blueprint for building a team science research network to understand the impact of the exposome—the measure of all environmental exposures over a lifetime and how those exposures relate to a person’s health—on autoimmune diseases. In FY 2023, NIH made several awards under this program, including projects focused on childhoodonset autoimmune diseases, minority health, and approaches to expand cohorts such as the NIH All of Us program to include exposome data. Ultimately, these efforts are expected to increase understanding of disease mechanisms, including mechanisms involving environmental factors, that influence rheumatic and autoimmune disease susceptibility, onset, and outcomes.
Progress in Skin Diseases
Hidradenitis suppurativa (HS) is a chronic, relapsing inflammatory disease of the skin, causing painful lumps to form in areas where the skin rubs together. Despite being relatively common, not much is known about the disease development of HS, leading to many challenges in diagnosis, symptom management, and treatment. In addition, NIH funding support for HS research was virtually absent until less than a decade ago. Recognizing this critical gap in knowledge, NIAMS has been providing fundamental support to researchers seeking to better understand the HS disease process and why HS disproportionally affects people, particularly women, of color. A NIAMS-supported research team performed a genome-wide association study in up to 720 people with HS and recently identified specific locations on the genome that contribute to one’s genetic risk of developing HS. Moving forward, the team will use the genetic basis of HS to inform new potential treatments for the disease. By learning more about how these recently discovered “risk locations” influence the HS disease process, they also may be able to apply their knowledge to better understand pain, inflammation, chronic wound formation, and health disparities in other inflammatory conditions.
On a related front, NIAMS recently announced the Institute’s special interest in supporting skin disease research to uncover the basis for suboptimal diagnosis, increased prevalence, elevated clinical severity, and altered treatment responses in understudied groups. Also, under a Notice of Special Interest on pain research, which is discussed further below, NIAMS funded research to better understand HS pain mechanisms, assess patients’ pain management needs, and begin to design ways to alleviate pain caused by HS.
Progress in Pain Research
Many diseases and conditions within the NIAMS mission areas cause pain, which affects people’s mobility and quality of life. Despite the frequency with which patients experience pain, the mechanisms underlying pain are not clearly understood. Furthermore, measures and treatment options that can prevent and alleviate pain caused by many types of arthritis and musculoskeletal and skin diseases remain a critical knowledge gap.
Credit: iStock
In FY 2023, NIAMS published two Notices of Special Interest focused on pain research in diseases and conditions within the Institute’s purview. NIAMS funded several pain research projects and six training projects that range from investigating new therapeutic pain relief without using opioids, to generation of a new model of pain research using skin organoids, and many others that will help to better understand the mechanisms of pain. Additionally, NIAMS is seeing progress in pain research programs that the Institute leads under the NIH-wide Helping to End Addiction Long-term® (HEAL) Initiative. One such program is the Back Pain Consortium (BACPAC) Research Program. Back pain is one of the most common medical problems in the United States and often is a contributing factor for opioid use disorder. The BACPAC Research Program is a patient-centered effort to develop effective and personalized therapies for chronic low back pain. For example, BACPAC funded a study that found a video camera developed for use in gaming systems could be used to differentiate patients with chronic low back pain into treatable subgroups. This commercially available video game camera could improve treatment by eliminating the need for complex measurement systems. In addition to BACPAC, NIAMS supports an ongoing clinical project analyzing racial differences in chronic low back pain. This study will be the first of its kind to examine psychological, socioeconomic, epigenomic, and transcriptomic data in a racially diverse group of adults with chronic low back pain.
Credit: iStock
Also, as part of HEAL, NIAMS leads the Restoring Joint Health and Function to Reduce Pain (RE-JOIN) Consortium to understand joint pain and enhance pain management. Projects address the knee, which is one of the most stressed joints in the body, and the temporomandibular (jaw) joint, which is one of the most understudied. It also supports two data management cores. By systematically mapping nerves in joints, RE-JOIN will help to develop targeted therapies to reduce pain and eliminate opioid dependency.
Looking to the future, NIAMS seeks to integrate BACPAC and RE-JOIN with other HEAL initiatives focused on pain into a whole joint health program with the goal of better understanding the shared mechanisms that influence pain across all tissues and joints. Through these collaborative efforts, we will strive to make significant advances in alleviating the chronic pain that afflicts our patients and society.
[1] pubmed.ncbi.nlm.nih.gov/37544309
IC Fact Sheet

Lindsey A. Criswell, M.D., M.P.H., D.Sc., became Director of NIAMS in February 2021. Previously, she served as vice chancellor of research at the University of California, San Francisco, as well as professor of rheumatology, and professor of orofacial sciences. Her research focuses on the genetics
and epidemiology of human autoimmune disease, particularly rheumatoid arthritis, and systemic lupus erythematosus. She has mentored many students and junior faculty to independent research careers.
History & Mission
The National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) was established in 1986. Its mission is to support research into the causes, treatment, and prevention of arthritis and musculoskeletal and skin diseases; the training of basic and clinical scientists to carry out this research; and the dissemination of information on research progress in these diseases.
Arthritis and musculoskeletal and skin conditions affect people of all ages and racial and ethnic backgrounds. Combined, they affect tens of millions of Americans, cause tremendous human suffering and disability, and cost the U.S. economy billions of dollars in health care costs and lost productivity. NIAMS aims both to foster an adaptable research environment that enables scientists to leverage opportunities as they arise and to stimulate new research in areas that are unexpected and transformative.
Recent Accomplishments
FDA recently approved beremagenegeperpavec-svdt as the first topical gene therapy for dystrophic epidermolysis bullosa (DEB), following years of NIAMS-funded research on DEB.
NIAMS intramural researchers found that dysregulation in neutrophil extracellular trap formation and degradation may contribute to COVID-19 disease progression and severity in patients. Additionally, NIAMS researchers reported improved clinical outcomes for hospitalized COVID-19 patients with treatment of fostamatinib—a drug used to treat low platelet levels.
NIAMS-funded basic research paved the way for recent FDA approval of delandistrogene moxeparvovec-rokl as the first genetic therapy for Duchenne muscular dystrophy. This recombinant gene therapy enables the production of part of the dystrophin protein that is present in normal skeletal muscle.
NIAMS maintains an annual cohort of diversity supplement scholars and mentors to foster the scholars’ trajectories toward independent research careers. NIAMS also participates in several NIH-wide initiatives for workforce diversity, equity, inclusion, and accessibility (DEIA).
The Institute supports an award under the Maximizing Opportunities for Scientific and Academic Independent Careers program to facilitate the transition of postdoctoral fellows of diverse backgrounds into tenure-track faculty positions.
Additionally, NIAMS supports several Administrative Supplements to Recognize Excellence in DEIA Mentorship in the biomedical sciences.

Research Highlights
In a NIAMS-supported project, researchers discovered that fibroblasts—a common cell type in connective tissues throughout the body—could play a key role in acne development, further highlighting the cells’ emerging significance in skin inflammation and immunity.
NIAMS-funded researchers developed an at-home urine test for lupus nephritis that can be read on a smartphone, allowing for easy monitoring of disease activity and response to therapies.
NIAMS provided instrumental support in the development of drugs called Janus kinase inhibitors whose use to combat various diseases within the NIAMS mission continues to progress to include other health conditions such as
COVID-19.
NIAMS-funded researchers discovered that statins may protect against knee osteoarthritis (OA) progression in patients with generalized and early-stage OA.

A NIAMS-supported ancillary study found Vitamin D ineffective against bone fracture risk, suggesting a need to reconsider the standard practice of vitamin D supplementation for fracture prevention.
Future Activities
NIAMS will focus on expanding the scope of the NIH’s Helping to End Addiction Long-term® (HEAL) Initiative to integrate more musculoskeletal components into research to prevent and treat the pain that often leads to opioid misuse and with the goal of restoring joint health.
As part of the HEAL Initiative, NIAMS will participate in the HEAL KIDS (Knowledge, Innovation and Discovery Studies) Pain program led by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. HEAL KIDS will help to address knowledge gaps in pediatric pain research to move the field forward.
NIAMS will participate in a new AMP® initiative in FY 2025 on the systems biology of inflammation, with a focus on developing a data science ecosystem.

Current Activities
NIAMS is collaborating with the NIH Office on Autoimmune Disease Research to establish the EXposome in Autoimmune Disease Collaborating Teams PLANning Awards (EXACT-PLAN). The exposome refers to the impact of environmental exposures over a lifetime and how those exposures relate to a person’s health. This plan will build a team science research network to improve understanding of the role of the exposome in various autoimmune diseases.
Planning for the implementation of the FY 2025 to 2029 Strategic Plan is underway. NIAMS hosted a meeting with over 160 participants to discuss the state of the science, scientific workforce, and cross-cutting themes to include in the strategic plan.

NIAMS is the clinical lead for the NIH’s Molecular Transducers of Physical Activity Consortium (MoTrPAC), which is developing a comprehensive map of exercise-initiated, molecular changes throughout the body to determine the mechanisms by which physical activity affects human health.
Photo Credits: Restoration of dystrophin (stained green) in Duchenne muscular dystrophy (Courtney Young, M.S., Melissa Spencer lab, University of California, Los Angeles); woman with broken wrist (iStock); and family playing soccer (iStock).
Major Changes in the Budget Request
Major changes by budget mechanism and/or budget activity detail are briefly described below. Note that there may be overlap between budget mechanism and activity detail, and these highlights will not sum to the total change for the FY 2025 President’s Budget request for NIAMS, which is $689.7 million, an increase of $2.1 million from the FY 2023 Final level. NIAMS continues to place a priority on support to early-stage investigators. Within this funding level, NIAMS will pursue its highest research priorities through strategic investments and careful stewardship of appropriated funds.
Research Project Grants (RPGs) (-$11.6 million; total $442.9 million):
NIAMS will support a total of 914 Research Project Grant (RPG) awards in FY 2025. Noncompeting awards will increase by $24.8 million. Competing RPGs will decrease by 62 awards and $26.9 million.
Other Research (+$3.1 million; total $32.4 million):
NIAMS will support a total of 215 Other Research awards in FY 2025. These increases are distributed across all programmatic areas.
Intramural Research (+$4.3 million; total $80.2 million):
NIAMS will increase funding for intramural research by 5.7%. These increases are distributed across all programmatic areas and basic, translational or clinical research to accommodate costs of employee salary and benefits increases and increases to centrally funded services.
Research Management and Support (+$2.8 million; total $41.4 million):
NIAMS will increase funding for research management and support by 7.3%. These increases will accommodate costs of employee salary and benefits increases and increases to centrally funded services.
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Budget Mechanism *
(Dollars in Thousands)
| Mechanism | FY2023 Final | FY 2024 CR | FY 2025 President's Budget | FY2025 +/- FY2023 | |||||
| Number | Amount | Number | Amount | Number | Amount | Number | Amount | ||
| Research Projects | |||||||||
Noncompeting | 720 | $314,625 | 716 | $331,964 | 686 | $339,460 | -34 | $24,835 | |
Administrative Supplements | (74) | $14,817 | (71) | $5,568 | (71) | $5,600 | -(3) | -$9,217 | |
| Competing: | |||||||||
Renewal | 28 | $14,532 | 24 | $12,231 | 21 | $10,788 | -7 | -$3,745 | |
New | 213 | $90,116 | 179 | $75,844 | 158 | $66,895 | -55 | -$23,221 | |
Supplements | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
| Subtotal, Competing | 241 | $104,648 | 203 | $88,075 | 179 | $77,682 | -62 | -$26,966 | |
| Subtotal, RPGs | 961 | $434,090 | 919 | $425,606 | 865 | $422,742 | -96 | -$11,348 | |
SBIR/STTR | 47 | $20,399 | 49 | $20,224 | 49 | $20,166 | 2 | -$233 | |
| Research Project Grants | 1,008 | $454,489 | 968 | $445,830 | 914 | $442,908 | -94 | -$11,580 | |
| Research Centers | |||||||||
Specialized/Comprehensive | 43 | $44,641 | 44 | $45,000 | 45 | $45,558 | 2 | $917 | |
Clinical Research | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
Biotechnology | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
Comparative Medicine | 0 | $50 | 0 | $50 | 0 | $50 | 0 | $0 | |
Research Centers in Minority Institutions | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
| Research Centers | 43 | $44,691 | 44 | $45,050 | 45 | $45,608 | 2 | $917 | |
| Other Research: | |||||||||
Research Careers | 192 | $25,669 | 191 | $27,800 | 191 | $28,356 | -1 | $2,687 | |
Cancer Education | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
Cooperative Clinical Research | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
Biomedical Research Support | 0 | $0 | 0 | $0 | 0 | $0 | 0 | $0 | |
Minority Biomedical Research Support | 0 | $269 | 0 | $271 | 0 | $273 | 0 | $4 | |
Other | 25 | $3,376 | 24 | $3,816 | 24 | $3,816 | -1 | $439 | |
| Other Research | 217 | $29,314 | 215 | $31,887 | 215 | $32,445 | -2 | $3,130 | |
| Total Research Grants | 1,268 | $528,494 | 1,227 | $522,767 | 1,174 | $520,961 | -94 | -$7,533 | |
| Ruth L Kirschstein Training Awards: | FTTPs | FTTPs | FTTPs | FTTPs | |||||
Individual Awards | 68 | $3,164 | 68 | $3,228 | 68 | $3,293 | 0 | $129 | |
Institutional Awards | 261 | $16,781 | 261 | $17,116 | 261 | $17,458 | 0 | $677 | |
| Total Research Training | 329 | $19,945 | 329 | $20,344 | 329 | $20,751 | 0 | $806 | |
Research & Develop. Contracts | 36 | $24,690 | 36 | $25,900 | 36 | $26,317 | 0 | $1,626 | |
SBIR/STTR (non-add) | (0) | ($221) | (0) | ($352) | (0) | ($357) | (0) | ($136) | |
Intramural Research | 130 | $75,876 | 133 | $76,148 | 139 | $80,227 | 9 | $4,352 | |
Res. Management & Support | 111 | $38,635 | 117 | $40,306 | 119 | $41,441 | 8 | $2,807 | |
SBIR/Admin. (non-add) | (0) | ($0) | (0) | ($0) | (0) | ($0) | (0) | ($0) | |
Construction | $0 | $0 | $0 | $0 | |||||
Buildings and Facilities | $0 | $0 | $0 | $0 | |||||
| Total, NIAMS | 241 | $687,639 | 250 | $685,465 | 258 | $689,697 | 17 | $2,058 | |
* All items in italics and brackets are non-add entries.
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
For carrying out section 301 and title IV of the PHS Act with respect to arthritis and musculoskeletal and skin diseases, $689,697,000.
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Summary of Changes
(Dollars in Thousands)
| CHANGES | FY 2023 Enacted | FY 2025 President's Budget | Built-In Change from FY 2023 Final | ||||||
| FTEs | Budget Authority | FTEs | Budget Authority | FTEs | Budget Authority | ||||
| 1. Intramural Research: | |||||||||
A. Built-in cost changes: | |||||||||
a. FY 2024 effect of FY 2023 pay & benefits increase | $27,211 | $31,251 | $321 | ||||||
b. FY 2024 effect of FY 2024 pay & benefits increase | $27,211 | $31,251 | 1,059 | ||||||
c. FY 2024 paid days adjustment | $27,211 | $31,251 | $105 | ||||||
d. Differences attributable to FY 2024 change in FTE | $27,211 | $31,251 | $558 | ||||||
e. FY 2025 effect of FY 2024 pay & benefits increase | $27,211 | $31,251 | $366 | ||||||
f. FY 2025 effect of FY 2025 pay & benefits increase | $27,211 | $31,251 | $502 | ||||||
g. FY 2025 paid days adjustment | $27,211 | $31,251 | $0 | ||||||
h. Differences attributable to FY 2025 change in FTE | $27,211 | $31,251 | $1,124 | ||||||
i. Payment for centrally furnished services | $11,745 | $12,593 | $849 | ||||||
j. Cost of laboratory supplies, materials, other expenses, and non-recurring costs | $36,905 | $36,383 | $2,848 | ||||||
Subtotal, IR built-in cost changes | $7,731 | ||||||||
2. Research Management and Support: | |||||||||
A. Built-in cost changes: | |||||||||
a. FY 2024 effect of FY 2023 pay & benefits increase | $21,264 | $24,061 | $251 | ||||||
b. FY 2024 effect of FY 2024 pay & benefits increase | $21,264 | $24,061 | $827 | ||||||
c. FY 2024 paid days adjustment | $21,264 | $24,061 | $82 | ||||||
d. Differences attributable to FY 2024 change in FTE | $21,264 | $24,061 | $577 | ||||||
e. FY 2025 effect of FY 2024 pay & benefits increase | $21,264 | $24,061 | $287 | ||||||
f. FY 2025 effect of FY 2025 pay & benefits increase | $21,264 | $24,061 | $389 | ||||||
g. FY 2025 paid days adjustment | $21,264 | $24,061 | $0 | ||||||
h. Differences attributable to FY 2025 change in FTE | $21,264 | $24,061 | $379 | ||||||
i. Payment for centrally furnished services | $2,851 | $3,056 | $206 | ||||||
j. Cost of laboratory supplies, materials, other expenses, and non-recurring costs | $14,475 | $14,324 | $1,073 | ||||||
Subtotal, RMS built-in cost changes | $4,071 | ||||||||
| CHANGES | FY 2023 Final | FY 2025 President's Budget | Program Change from FY 2023 Final | ||||||
| No. | Amount | No. | Amount | No. | Amount | ||||
| B. Program: | |||||||||
1. Research Project Grants: | |||||||||
a. Noncompeting | 720 | $329,442 | 686 | $345,060 | -34 | $15,618 | |||
b. Competing | 241 | $104,648 | 179 | $77,682 | -62 | -$26,966 | |||
c. SBIR/STTR | 47 | $20,399 | 49 | $20,166 | 2 | -$233 | |||
Subtotal, RPGs | 1,008 | $454,489 | 914 | $442,908 | -94 | -$11,580 | |||
2. Research Centers | 43 | $44,691 | 45 | $45,608 | 2 | $917 | |||
3. Other Research | 217 | $29,314 | 215 | $32,445 | -2 | $3,10 | |||
4. Research Training | 329 | $19,945 | 329 | $20,751 | 0 | $806 | |||
5. Research and development contracts | 36 | $24,690 | 36 | $26,317 | 0 | $1626 | |||
| Subtotal, Extramural | $573,129 | $568,029 | -$5,100 | ||||||
6. Intramural Research | 130 | $75,876 | 139 | $80,227 | 9 | -$3,380 | |||
7. Research Management and Support | 111 | $38,635 | 119 | $41,441 | 8 | -$1,265 | |||
8. Construction | $0 | $0 | $0 | ||||||
9. Buildings and Facilities | $0 | $0 | $0 | ||||||
Subtotal, Program | -$9,745 | ||||||||
Total built-in and program changes | 241 | $687,639 | 258 | $689,697 | 17 | $2,058 | |||
Budget Graphs
History of Budget Authority and FTEs:
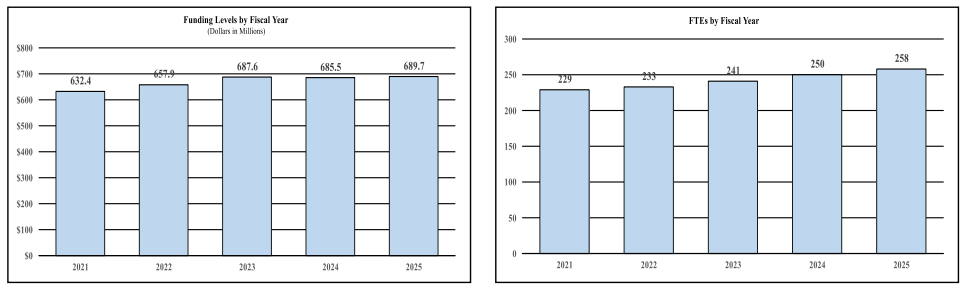
Distribution by Mechanism:

Change by Selected Mechanisms:

NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Organizational Chart
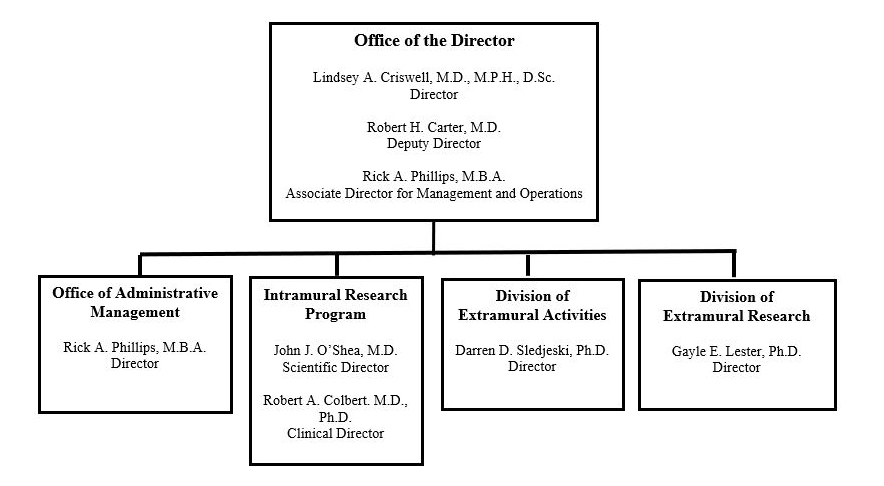
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Budget Authority by Activity*
(Dollars in Thousands)
| FY 2023 Final | FY 2024 CR | FY 2025 President's Budget | FY 2025 +/- FY 2023 Final | |||||||||
| Extramural Research |
|
|
|
| ||||||||
| Detail | ||||||||||||
| Systemic Rheumatic and Autoimmune Diseases | $95,598 | $94,912 | $94,748 | -$851 | ||||||||
| Skin Biology and Diseases | $117,400 | $116,556 | $116,355 | -$1,045 | ||||||||
| Muscle Biology and Diseases | $87,938 | $87,306 | $87,156 | -$783 | ||||||||
| Joint Biology and Diseases and Orthopaedics | $194,363 | $192,966 | $192,633 | -$1,730 | ||||||||
| Bone Biology and Diseases | $77,830 | $77,270 | $77,137 | -$693 | ||||||||
Subtotal, Extramural | $573,129 | $569,011 | $568,029 | -$5,100 | ||||||||
| Intramural Research |
|
|
|
| ||||||||
| Research Management & Support |
|
|
|
| ||||||||
| TOTAL |
|
|
|
|
* Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
Justification of Budget Request
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Authorizing Legislation: Section 301 and Title IV of the Public Health Service Act, as amended.
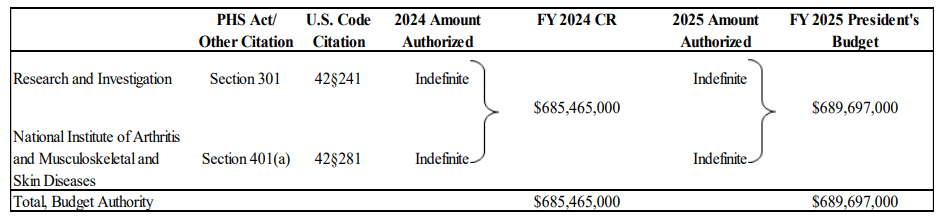
Budget Authority (BA):
| FY 2023 Final | FY 2024 Continuing Resolution | FY 2025 President's Budget | FY 2025+/- FY 2023 | |
| BA FTE | $687,639,000 241 | $685,465,000 250 | $689,697,000 258 | $2,058,000 +17 |
Program funds are allocated as follows: Competitive Grants/Cooperative Agreements; Contracts; Direct Federal/Intramural and Other.
Overall Budget Policy:
FY 2025 President’s Budget request for NIAMS is $689.7 million, an increase of $2.1 million or 0.3% compared with the FY 2023 Final level. This funding level will support basic, translational, and clinical research across all of NIAMS’s mission areas, as described below.
Program Descriptions
Systemic Rheumatic and Autoimmune Diseases
Research in this program seeks to understand the causes of rheumatic and autoimmune diseases and works to develop new approaches to management, treatment, and prevention. For example, researchers supported by NIAMS under this program recently discovered a molecular link between rheumatoid arthritis (RA) and periodontal (gum) diseases. People with RA who have periodontal diseases often show more severe RA symptoms, and this research documents a direct immune response leading to RA flares when the lining of the mouth is injured.
The autoimmune disease systemic lupus erythematosus (SLE) is also a key focus of this program. Researchers found that two specific immune system molecules, known as autoantibodies because they react with the body’s own proteins, impact SLE severity. These findings shed light on the autoantibodies that characterize SLE and may help identify patients who need early and aggressive treatment. Other NIAMS-funded researchers investigating the mechanisms by which inflammatory autoimmune B cells (ABCs) drive SLE pinpointed a specific receptor that influences inflammation caused by the cells. This has therapeutic implications for several other autoimmune diseases within the NIAMS portfolio because ABCs are found in Sjögren’s disease, RA, Crohn’s disease, multiple sclerosis, and scleroderma.
Credit: iStock
NIAMS also funds the largest genome-wide association study of Sjögren’s disease to date. This international collaboration provided data from over 20,000 people that nearly doubles the number of known genetic regions associated with an increased risk of Sjögren’s disease. NIAMS-funded researchers also analyzed the prevalence of gout by examining sex and racespecific disparities among adults in the United States. They found that Black men and women are at the highest risk of gout, which challenges the historical notion that gout predominantly impacts affluent White men. Social circumstances such as poverty and certain lifestyle factors were also found to be risk factors for gout.
Budget Policy:
The FY 2025 President’s Budget for this program is $94.7 million, a decrease of $0.9 million or 0.9 percent compared with the FY 2023 Final level. In FY 2025, NIAMS will continue supporting the Accelerating Medicines Partnership Autoimmune and Immune-Mediated Diseases program (AMP AIM) to identify cellular and molecular mechanisms that impact inflammation in a variety of autoimmune and immune-mediated diseases. NIAMS also will fund the Arthritis and Autoimmune and Related Diseases Knowledge (ARK) Portal. In FY 2025, NIAMS will participate in a new AMP program on the Systems Biology of Inflammation. The program will leverage multi-omics datasets to identify shared molecular pathways of inflammation across diseases.
Skin Biology and Diseases

Credit: Adobe Stock
The Skin Biology and Diseases program supports research on the properties and functions of healthy skin, the causes of skin disease, and new approaches for skin disease prevention and treatment. For example, the program portrait on gene therapy describes a recent advance in gene therapy for epidermolysis bullosa, a genetic skin condition, that built on years of NIAMS- funded advances. In addition, research supported by the program seeks to better understand the biologic mechanisms of the hair growth cycle, including hair graying in older people. A better understanding of how stem cells are regulated during hair growth to properly maintain the human body and replace tissues damaged by injuries is of critical importance because stem cell dysregulation can cause many health problems, including cancer. A team of NIAMS-funded researchers found that the stem cells responsible for hair color can switch between stem cell and mature states, depending on whether they live in a part of the hair follicle known as the bulge or an area called the hair germ. With age, these cells can get stuck between the bulge and the hair germ and cannot become mature melanocytes for pigmentation or maintain their stem cell state for later rounds of hair pigmentation. These findings suggest that these stuck cells may be responsible for graying and loss of hair color and, if their movement can be restored, it might prevent or reverse hair graying.
Congenital giant nevi are huge, pigmented moles that are typically present at birth and may cover much of the face and body. Due to the moles’ appearance and their risk of later developing into skin cancer, many parents decide to have their children undergo extensive surgery to remove the entire lesion, which can cause large and permanent scars. NIAMS-funded researchers recently created multiple animal models of this skin condition
and used them to show that several drugs can be applied to the skin to cause the lesions to regress. They also found one topical drug that protects against skin cancer. These findings help set the stage for future studies of safety, potential enhancements of efficacy, and more analysis of underlying mechanisms to treat congenital giant nevi and prevent them from developing into skin cancer.

Credit: iStock
Finally, this program also supports research on chronic itch, which occurs in many medical conditions and in response to certain drugs. Chronic itch affects millions of Americans, yet its causes are poorly understood. A NIAMS-supported team investigated contagious itch behavior, a phenomenon known for decades, if not centuries. The researchers found that in mice, this so-called “contagious itching” is controlled through a visual pathway that operates independently of the visual cortex, the area of the brain that processes visual information. Instead, the conversion of visual information from seeing an adjacent mouse scratching to an itch sensation in one’s own body appears to be mediated by a nerve fiber that originates in the retina and ends in a tiny brain region responsible for controlling circadian rhythm, triggering activation of the scratching cascade. Understanding this pathway will have a multifaceted impact on cross-sensory signal processing, communication, and behavior modulation, which may lead to novel sensory interventions to treat itch, pain, and other disorders.
Program Portrait: Gene Therapy
The FDA recently approved beremagene geperapvec-svdt, a herpes-simplex virus type 1 (HSV.1), vector-based gene therapy for the treatment of wounds in patients with dystrophic epidermolysis bullosa (DEB), a genetic disorder that affects the connective tissues in the skin and nails. The treatment is specifically for DEB patients six months of age and older with mutations in the collagen type VII alpha 1 chain (COL7A1) gene. The genetic mutations can cause painful and debilitating blisters and wounds. DEB usually presents itself at birth and is divided into two major types depending on the inheritance pattern: recessive dystrophic epidermolysis bullosa and dominant dystrophic epidermolysis bullosa. NIAMS and other NIH components funded early work on various gene therapy models for DEB.
In addition, the FDA recently granted accelerated approval for the treatment of Duchenne muscular dystrophy (DMD) patients between the ages of 4 and 5 years with delandistrogene moxeparvovec. This is the first gene therapy for DMD, a rare genetic disorder characterized by progressive muscle weakness and loss of function due to mutations in the dystrophin gene. Research funded by NIH laid the groundwork by identifying gene therapy as a promising potential treatment strategy for DMD. NIH funded the foundational efforts in DMD gene therapy at some of the Paul D. Wellstone Muscular Dystrophy Specialized Research Centers, which spurred early research in DMD. Gene therapy aims to address DMD by delivering a functional copy of dystrophin into skeletal muscle cells. The company Sarepta built on the previous work to encode a much smaller protein called microdystrophin that exerts its therapeutic effect by partially reproducing the functionality of full-length dystrophin.
Budget Policy:
The FY 2025 President’s Budget request for this program is $116.4 million, a decrease of $1.0 million or 0.9 percent compared with the FY 2023 Final level. Program plans for FY 2025 include continued support for the NIAMS Skin Biology and Diseases Resource-based Centers. These centers provide critical research infrastructure, shared facilities, services, and resources to groups of investigators studying skin biology and diseases, enabling them to conduct their independently funded individual or collaborative research projects more efficiently and effectively. The program also plans to continue support for research on skin diseases and conditions in racial and ethnic groups where these diseases and conditions have been understudied. This work addresses a fundamental research gap that has created a significant unmet need for better prevention and improved therapeutic approaches in diverse populations.
Muscle Biology and Diseases
This program supports studies to explain the muscle’s role in health and disease, leading to new therapeutic treatment and prevention of skeletal muscle diseases and disorders. This program is one of many at NIAMS focused on health at all stages of life and on both common and rare diseases. For example, a group of researchers studying Duchenne muscular dystrophy (DMD) in a mouse model compared the drug prednisolone to spironolactone for 1 week at 5.5 weeks of life. They observed differences in inflammatory markers and levels of infiltrating immune cells in a skeletal muscle model and in the diaphragm between prednisolone-treated and spironolactone-treated mice.
In another lab, NIAMS-supported researchers studied the overexpression of the protein sarcospan in muscle cells of dystrophic mice and found that it restores the mechanical link between muscle cells and their surroundings, known as the extracellular matrix (ECM), and protects the muscle from injury. By comparing RNA and protein expression in muscles from control, dystrophic, and dystrophic mice with overexpression of sarcospan, researchers determined that increased sarcospan results in compensatory changes in the composition of the ECM and cytoskeleton in dystrophic muscles. The researchers suggest that interventions targeting regulators of mechanical signaling cascades could improve function in dystrophic muscles of patients with Duchenne muscular dystrophy. In addition to the studies described here, information about approval of a gene therapy for DMD is described in the gene therapy program portrait.
The NIAMS muscle research portfolio also emphasizes research in cachexia, which is a common cancer-related multi-organ syndrome accompanied by muscle wasting. Currently, there is no treatment for cachexia. A research group found a mechanism that leads specifically to muscle fiber degradation in mouse models of human breast cancer. The mechanism involves calcium accumulation within the muscle cells corresponding to activation of enzymes that break down proteins and subsequent cleavage of filaments, resulting in the destruction of muscle fibers. In another study, researchers suggested that damage to skeletal muscle mitochondria contributes to the development of cachexia. Using a mouse cancer model, the researchers measured weakness, atrophy, and mitochondrial energy response in the quadriceps and diaphragm during cancer progression. They found that in both muscles, weakness preceded atrophy, but alterations in the muscles’ mitochondrial functions differed.
Budget Policy:
The FY 2025 President’s Budget request for this program is $87.2 million, a decrease of $0.8 million or 0.9 percent compared with the FY 2023 Final level. NIAMS plans for FY 2025 include continued support for the Paul D. Wellstone Muscular Dystrophy Research Centers, which promote collaborative basic, translational, and clinical research, and provide important resources for muscular dystrophy researchers nationwide. One center, funded through 2026, is organized around the central theme of preserving skeletal and cardiac muscle function in muscular dystrophies by delineating disease-modifying targets and developing therapeutic strategies.
Joint Biology and Diseases and Orthopaedics
The Joint Biology and Diseases and Orthopaedics program focuses on the interplay among the body’s muscles, bones, and connective tissues. This program supports basic, translational, and clinical research centered on the dynamics among these different aspects of the body. Joint injuries often require painful rehabilitation and can lead to prolonged disability. Many ongoing projects aim to regenerate the crucial elements of a joint, ideally by reprograming some of the patient’s own stem cells. Researchers developed a method to generate tendon cells from induced pluripotent stem cells (iPSCs)—a type of cell that can be reprogrammed to be capable of dividing and developing into the three primary groups of cells that make up a human body. This method establishes a way to generate an unlimited number of these cells for tendon repair. Results from this study have the potential to significantly advance regenerative approaches to treating tendon injuries. Similar methods of using iPSCs were also found to generate cartilage that can then be turned into ligament cells for ligament injury repair.

Credit: iStock
Osteoarthritis (OA) is the most common joint disease and a leading cause of activity limitation, yet current treatments are only partially effective. Recently, FDA approved a treatment promoting anterior cruciate ligament (ACL) healing; more information is in the program portrait for BEAR-MOON. In addition, NIAMS-funded researchers discovered a specific molecular pathway that occurs in the synovial membrane of joints, an area that when injured, leads to post-traumatic OA. These findings suggest a novel pathway underlying the disease, expanding the knowledge of disease onset. In another NIAMS-funded project, researchers found that statins, a family of drugs widely used in cardiovascular medicine, may protect against knee OA progression in patients with generalized or early-stage OA.

Credit: iStock
Additionally, people with knee OA often have limited physical activity and walking endurance, even after total knee replacement (TKR). This may lead to increased cardiovascular events such as heart attacks and strokes, and early death. Functional assessments are critical measures to inform the care and treatment of patients with knee OA, but their relationship to everyday function has not been well studied. Results from a NIAMS-funded study suggest that by improving walking endurance, people can improve physical activity and quality of life post-TKR. These results support the need for walking-based rehabilitation to improve physical function and quality of life after TKR, and probably after other knee-related surgeries. Another NIAMS-supported research team showed that wearable, sensor-based measures can be used to reliably assess gait and sit-to-stand ability in individuals' homes. This low-cost and reliable tool can be used in future clinical trials and studies to improve patient care for people with knee OA and other musculoskeletal issues.
Other NIAMS-funded researchers have focused on the spine and found that the previous method of spine alignment imaging, where patients are sitting upright for imaging and lying face up during surgery, does not produce the best clinical outcomes. Instead, researchers suggest imaging in a face-up position that more accurately mimics patients’ anatomy during surgery, which will allow surgeons to be more precise when performing a procedure.
Program Portrait: BEAR-MOON
In December 2020, the U.S. Food and Drug Administration (FDA) approved the first-ever implant to stimulate anterior cruciate ligament (ACL) healing. Approximately 20,000 ACL reconstructions take place every year, but this process can be very invasive and may not restore joint motion. Further, many people cannot return to the same level of activity following the surgery. To address these issues, researchers began to investigate how to restore the ACL rather than repair it and keep muscles and tendons around the knee intact. This research resulted in the NIAMS-funded development of the first of its kind, bridge-enhanced ACL restoration (BEAR) implant.
The BEAR device uses structural cells from the extracellular matrix to hold blood in the ACL tear site, promoting a blood clot and eventual healing. Several clinical trials followed the development of this device proving its safety and feasibility, leading to the FDA approval. At the same time, a NIAMS-supported clinical trial called BEAR-MOON was established to compare the results of the BEAR implant to traditional reconstructive surgery using the Multicenter Orthopaedic Outcomes Network, an effort designed to study ACL reconstruction outcomes. This trial has continued beyond FDA approval of the device to generate further data about its long-term effects.
NIAMS-supported researchers are also training orthopaedic surgeons to conduct the implant procedure so that more people can benefit. The BEAR-MOON trial has also shifted focus to highly active patients who are at a greater risk of ACL re-tear due to lifestyle post-surgery. As this device continues to be used, other NIAMS-funded researchers are looking into post-operative care and its impact on the risk of re-injury.
Budget Policy:
The FY 2025 President’s Budget request for this program is $192.6 million, a decrease of $1.7 million or 0.9 percent compared with the FY 2023 Final level. In FY 2025, NIAMS will continue supporting resource-based centers for bone, muscle, and orthopaedic research. This funding will provide infrastructure, facilities, services, and resources to investigators focused on joint and orthopaedic health. NIAMS will also support Infrastructure for Musculoskeletal Pediatric Acute Care Clinical Trials (IMPACCT), a grant to develop the infrastructure necessary for multicenter randomized clinical trials in pediatric fractures. This framework will help guide clinical decision-making for managing these injuries.
Bone Biology and Diseases
This program supports scientific advances that benefit the foundational core of human health. Project areas range from bone disease, both common and rare, to fundamental bone biology and its translational applications. Millions of Americans have osteoporosis, a debilitating and potentially life threatening condition in which bones become weak and brittle. NIAMS-funded researchers demonstrated that a single dose of zoledronic acid, a commonly prescribed osteoporosis treatment, is sufficient to maintain certain benefits brought about by combinations of other treatments. However, the compound itself does not improve bone strength and microarchitecture. These findings may lead to increases in treatment adherence and improvements in treatment outcomes.

Credit: Frederick Kaplan, M.D.
Research funded by NIAMS is also bringing cures into reach for the rare condition fibrodysplasia ossificans progressiva (FOP) while also uncovering clues into fundamental biology. In people with FOP, bone replaces muscle and connective tissue. Extensive research funded by NIAMS led to the development of the first FOP drug treatment to be FDA-approved. The drug, palvarotene, received FDA approval in August 2023. Other FOP research has revealed that the condition is primarily caused by a genetic mutation affecting a member of a family of molecules involved in many biologic processes. NIAMS-funded researchers focusing on the mutation have shown that overexpression of the same gene that is free of the mutation could be the basis of a new highly effective, safe FOP treatment. Their research has also revealed a rare biologic phenomenon that can provide insight into complicated biology far beyond FOP.
Numerous program findings have applications for and beyond bone health, including advances related to obesity and circadian rhythms. One study shed light on the interrelationship between metabolism and bone biology, identifying a target for both obesity and osteoporosis treatment. In a separate study, researchers discovered a molecular pathway by which melatonin, a popular dietary supplement hormone, affects bone, with effects differing between female and male mice. The results of this study can be applied to research related to the health effects of sleep disruptions, aging, and menopause.
Budget Policy:
The FY 2025 President’s Budget request for this program is $77.1 million, a decrease of $0.7 million or 0.9 percent compared with the FY 2023 Final level. This program is leading the agency’s efforts towards the Appropriate Use of Drug Therapies for Osteoporotic Fracture Prevention as described in the 2019 Pathways to Prevention workshop of the same name. NIAMS, in collaboration with other Institutes and Centers, is addressing the recommendations from the workshop and will continue to do so in FY 2025. NIAMS plans to continue support for the pooling of international cohort studies of treatments and fractures to develop risk prediction models enabling individualized care that accounts for personalized risks and benefits of osteoporosis therapy.
Intramural Research Program
Researchers in the NIAMS Intramural Research Program (IRP) study the genetics, etiology, pathogenesis, and treatment of rheumatic, autoimmune, inflammatory, bone, skin, and muscle diseases. In a recent study, NIAMS intramural researchers investigated the relationship between how cells called neutrophils infiltrate into the synovial joint—a hallmark of RA—and the disease’s characteristic bone erosion. They found that neutrophil extracellular traps (NETs), a netlike structure released by neutrophils, play a direct role in promoting progressive bone erosion and the formation of osteoclasts—the cells that degrade bone—in people with RA. Their finding suggests that targeting NET formation might prevent RA-associated bone damage and mitigate its debilitating effects.
Another team of NIAMS intramural researchers discovered that NETs also are involved in diabetic foot ulcers (DFUs), a life-threatening complication of diabetes mellitus. Unlike typical injuries in people without diabetes, DFUs often stubbornly refuse to heal, frequently get infected, and sometimes require amputation of the affected limb. The researchers found that the absence of a protein called FOXM1 in neutrophils leads to excessive NET formation and impairs wound healing, whereas the presence of a protein called TREM1 predicts normal wound healing. These findings indicate that the interplay between TREM1, FOXM1, and NET production influences healing. A better understanding of that relationship may reveal therapeutic strategies to promote DFU healing.
Further, NIAMS intramural researchers and their collaborators at the National Human Genome Research Institute surveyed the skin microbiome of patients with atopic dermatitis (AD, also called eczema) to understand the genetic diversity of the bacteria present on the skin and how genetic variants may contribute to disease severity. They identified specific staphylococcal bacteria strains and genes associated with disease presence and severity. Their results provide information about the vast diversity of skin microbes in AD, which could help inform efforts to identify potential new therapeutics for preventing or treating severe disease.
Program Portrait: The Hope Center
The NIAMS Intramural Research Program (IRP) is actively engaged in clinical research, using the expertise of its scientists and the resources of the NIH
Clinical Center to better diagnose and treat both common and rare rheumatic diseases. To aid in the IRP’s research and training mission, in 2001, the
NIAMS Community Health Clinic (CHC) was established as part of its Clinical Care and Research effort. The CHC’s goals are to better understand diseases such as rheumatoid arthritis and lupus and to gain insights about why these and other rheumatic diseases disproportionately affect women and people of color. Based at the NIH Clinical Center in Bethesda, Maryland, the CHC is a clinical research site where NIAMS researchers provide rheumatology specialty care; gather health information to answer research questions about rheumatic diseases in certain groups of people; and provide scientifically based health information on rheumatic, musculoskeletal, and skin diseases.
NIAMS medical experts in the fields of autoimmunity, and rheumatologic diseases also conduct research focused on the evaluation, diagnosis, standard of care, and disease management of underserved communities. To that end, in collaboration with the National Heart, Lung, and Blood Institute (NHLBI), NIAMS has re-engaged with the Washington, D.C., community through the Hope Center. The Institute’s participation in the Hope Center operates under the direction of the CHC and helps facilitate the NIAMS IRP’s efforts to reach underserved populations in the local area who are living with rheumatic diseases and who qualify to participate in research protocols. In 2023, the NIAMS intramural researchers set up a team at the Washington, D.C.-based, community focused research facility that they share with collaborators, including their NHLBI colleagues, to conduct outreach and allow participants in clinical trials to be seen close to their homes. By becoming more involved with underserved communities, the researchers hope to better understand the challenges they face, which, in turn, can better inform NIAMS’s research and responses. As the vast disparities in health care and health care access affect groups that have historically and continually faced increased barriers to, or exclusion from, many aspects of life, the NIAMS IRP is committed to direct and meaningful engagement with communities to ultimately reduce or eliminate these disparities and allow for a future where every person has access to needed health care.
Researchers within the NIAMS IRP conduct patient-based investigations in rheumatic, skin, muscular, and inflammatory diseases and train the next generation of clinician-investigators in these areas. For example, in a recent clinical study, they found that pioglitazone, a drug used to treat type 2 diabetes, is well tolerated, and induces significant improvement in vascular stiffness and cardiometabolic parameters associated with premature cardiovascular disease in people with SLE. Another study was conducted to test the effect of abatacept—a biologic treatment for rheumatoid arthritis—in juvenile dermatomyositis (JDM). JDM is an autoimmune disease in children that causes skin rash and muscle inflammation resulting in weak muscles. The researchers found that abatacept is safe and beneficial to children with treatment-resistant JDM.
Finally, the NIAMS IRP continues to explore areas of opportunity for its innovative research. In FY 2023, it co-sponsored a meeting to identify unmet needs in spondyloarthritis focusing on disease mechanisms, social constructs, and health disparities, as well as pain and therapeutics.
Budget Policy:
The FY 2025 President’s Budget request for this program is $80.2 million, an increase of $4.4 million or 5.7 percent compared with the FY 2023 Final level. Program plans for FY 2025 include continuing efforts in genetic sequencing and spatial transcriptomics, expanding capabilities in computational biology, and optimizing light imaging technology to drive scientific discovery. The program also will continue its momentum in using genomic sequencing and genetic counseling services to help identify the genetic causes of childhood lupus. In addition, the NIAMS IRP is committed to implementing policy and guidance in compliance with the NIH 2023 Data Management and Sharing Policy. Finally, the program will continue its progress in improving stipends provided to its research trainees at all levels.
Research Management and Support (RMS)
The RMS budget supports the scientific, administrative management, and information technology activities associated with NIAMS’ day-to-day operations. In FY 2023, NIAMS managed 1,268 research grants and centers, as well as 36 research and development contracts and 329 individual and institutional full-time research training positions. NIAMS supported 599 clinical research studies, including 109 clinical trials. In FY 2023, NIAMS invited more than 160 researchers and community members to provide input on the themes for the FY 2025 to 2029 strategic plan. In FY 2024, NIAMS organized workshops for the regenerative medicine innovation project (RMIP) to hear investigators’ insights about new directions in this area of medicine.
Budget Policy:
The FY 2025 President’s Budget request for this program is $41.4 million, an increase of $2.8 million or 7.3 percent compared with the FY 2023 Final level. Program plans for FY 2025 include the implementation of the Institute’s strategic plan for FY 2025-2029 and support of the Muscular Dystrophy Coordinating Committee (MDCC) development of a new action plan.
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Appropriations History
Research Management and Support (RMS)
| Fiscal Year | Budget Estimate to Congress | House Allowance | Senate Allowance | Appropriation |
| 2016 Rescission | $533,232,000 | $528,137,000 | $544,274,000 | $542,141,000 $0 |
| 20171 Rescission | $541,662,000 | $555,181,000 | $564,131,000 | $557,851,000 $0 |
| 2018 Rescission | $417,898,000 | $566,515,000 | $576,178,000 | $586,661,000 $0 |
| 2019 Rescission | $545,494,000 | $593,663,000 | $605,383,000 | $605,065,000 $0 |
| 2020 Rescission | $520,829,000 | $634,637,000 | $637,097,000 | $624,889,000 $0 |
| 2021 Rescission | $568,480,000 | $635,263,000 | $645,237,000 | $634,292,000 $0 |
| 2022 Rescission | $680,186,000 | $679,410,000 | $675,106,000 | $655,699,000 $0 |
| 2023 Rescission | $676,254,000 | $676,395,000 | $686,025,000 | $685,465,000 $0 |
| 2024 Rescission | $687,639,000 | $685,465,000 | $685,465,000 | $685,465,000 $0 |
| 2025 | $689,697,000 |
1 Budget Estimate to Congress includes mandatory financing.
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Authorizing Legislation
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Amounts Available for Obligation¹
(Dollars in Thousands)
| Source of Funding | FY 2023 Final | FY 2024 CR | FY 2025 President's Budget |
| Appropriation | $685,465 | $685,465 | $689,697 |
| Mandatory Appropriation: (non-add) | |||
Type 1 Diabetes | ($0) | ($0) | ($0) |
Other Mandatory financing | ($0) | ($0) | ($0) |
| Subtotal, adjusted appropriation | $685,465 | $685,465 | $689,697 |
| OAR HIV/AIDS Transfers | $2,174 | $0 | $0 |
| Subtotal, adjusted budget authority | $687,639 | $685, 465 | $689, 697 |
| Unobligated balance, start of year | $0 | $0 | $0 |
| Unobligated balance, end of year (carryover) | $0 | $0 | $0 |
| Subtotal, adjusted budget authority | $687,639 | $685,465 | $689,697 |
| Unobligated balance lapsing | -$60 | $0 | $0 |
| Total obligations | $687,579 | $685,465 | $689,697 |
¹ Excludes the following amounts (in thousands) for reimbursable activities carried out by this account:
- FY 2023 - $4,460
- FY 2024 - $4,681
- FY 2024 - $4,813
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Budget Authority by Object Class¹
(Dollars in Thousands)
| FY 2024 CR | FY 2025 President's Budget | |
| Total compensable work years: | ||
Full-time equivalent | 250 | 258 |
Full-time equivalent of overtime and holiday hours | 1 | 1 |
Average ES salary | $197 | $201 |
Average GM/GS grade | 13.0 | 13.0 |
Average GM/GS salary | $142 | $145 |
Average salary, Commissioned Corps (42 U.S.C. 207) | $114 | $119 |
Average salary of ungraded positions | $172 | $175 |
| OBJECT CLASSES | FY 2024 CR | FY 2025 President's Budget |
Personnel Compensation | ||
| 11.1 Full-Time Permanent | $21,964 | $23,215 |
| 11.3 Other Than Full-Time Permanent | $11,357 | $12,108 |
| 11.5 Other Personnel Compensation | $1,537 | $1,617 |
| 11.7 Military Personnel | $481 | $503 |
| 11.8 Special Personnel Services Payments | $3,986 | $4,097 |
| 11.9 Subtotal Personnel Compensation | $39,361 | $41,540 |
| 12.1 Civilian Personnel Benefits | $12,815 | $13,679 |
| 12.2 Military Personnel Benefits | $88 | $92 |
| 13.0 Benefits to Former Personnel | $0 | $0 |
| Subtotal Pay Costs | $52,264 | $55,312 |
| 21.0 Travel & Transportation of Persons | $546 | $558 |
| 22.0 Transportation of Things | $179 | $183 |
| 23.1 Rental Payments to GSA | $0 | $0 |
| 23.2 Rental Payments to Others | $6 | $6 |
| 23.3 Communications, Utilities & Misc. Charges | $78 | $80 |
| 24.0 Printing & Reproduction | $9 | $10 |
| 25.1 Consulting Services | $20,648 | $21,287 |
| 25.2 Other Services | $14,941 | $15,264 |
| 25.3 Purchase of Goods and Services from Government Accounts | $47,360 | $48,502 |
| 25.4 Operation & Maintenance of Facilities | $133 | $136 |
| 25.5 R&D Contracts | $3,913 | $3,875 |
| 25.6 Medical Care | $140 | $145 |
| 25.7 Operation & Maintenance of Equipment | $4,167 | $4,324 |
| 25.8 Subsistence & Support of Persons | $0 | $0 |
| 25.0 Subtotal Other Contractual Services | $91,301 | $93,533 |
| 26.0 Supplies & Materials | $4,053 | $4,303 |
| 31.0 Equipment | $2,314 | $2,405 |
| 32.0 Land and Structures | $0 | $0 |
| 33.0 Investments and Loans | $0 | $0 |
| 41.0 Grants, Subsidies & Contributions | $534,704 | $533,297 |
| 42.0 Insurance Claims & Indemnities | $0 | $0 |
| 43.0 Interest & Dividends | $10 | $10 |
| 44.0 Refunds | $0 | $0 |
Subtotal Non-Pay Costs | $633,201 | $634,385 |
Total Budget Authority by Object Class | $685,465 | $689,697 |
1 Includes FTEs whose payroll obligations are supported by the NIH Common Fund.
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Salaries and Expenses
(Dollars in Thousands)
| Object Classes | FY 2024 CR | FY 2025 President's Budget |
| Personnel Compensation | ||
Full-Time Permanent (11.1) | $21,964 | $23,215 |
Other Than Full-Time Permanent (11.3) | $11,357 | $12,108 |
Other Personnel Compensation (11.5) | $1,573 | $1,617 |
Military Personnel (11.7) | $481 | $503 |
Special Personnel Services Payments (11.8) | $3,986 | $4,097 |
Subtotal, Personnel Compensation (11.9) | $39,361 | $41,540 |
Civilian Personnel Benefits (12.1) | $12,815 | $13,679 |
Military Personnel Benefits (12.2) | $88 | $92 |
Benefits to Former Personnel (13.0) | $0 | $0 |
Subtotal Pay Costs | $52,264 | $55,312 |
Travel & Transportation of Persons (21.0) | $546 | $558 |
Transportation of Things (22.0) | $179 | $183 |
Rental Payments to Others (23.2) | $6 | $6 |
Communications, Utilities & Misc. Charges (23.3) | $78 | $78 |
Printing & Reproduction (24.0) | $9 | $10 |
| Other Contractual Services | ||
Consultant Services (25.1) | $20,174 | $20,802 |
Other Services (25.2) | $14,941 | $15,264 |
Purchase of Goods and Services from Government Accounts (25.3) | $28,809 | $29,815 |
Operation & Maintenance of Facilities (25.4) | $133 | $136 |
Operation & Maintenance of Equipment (25.7) | $4,167 | $4,324 |
Subsistence & Support of Persons (25.8) | $0 | $0 |
Subtotal Other Contractual Services | $68,223 | $70,341 |
| Supplies & Materials (26.0) | $4,053 | $4,303 |
| Subtotal Non-Pay Costs | $73,095 | $75,480 |
Total Administrative Costs | $125,360 | $130,792 |
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Detail of Full-Time Equivalent Employment (FTE)
| Office | FY 2023 Final | FY 2024 CR | FY 2025 President's Budget | |||||||||||
| Civilian | Military | Total | Civilian | Military | Total | Civilian | Military | Total | ||||||
| Division of Extramural Research | ||||||||||||||
| Direct | 29 | - | 29 | 31 | - | 31 | 31 | - | 31 | |||||
Total: | 29 | - | 29 | 31 | - | 31 | 31 | - | 31 | |||||
| Office of the Director | ||||||||||||||
| Direct | 59 | - | 59 | 61 | - | 61 | 62 | - | 62 | |||||
Total: | 59 | - | 59 | 61 | - | 61 | 62 | - | 62 | |||||
| Division of Extramural Research | ||||||||||||||
| Direct | 23 | - | 23 | 25 | - | 25 | 26 | - | 26 | |||||
Total: | 23 | - | 23 | 25 | - | 25 | 26 | - | 26 | |||||
| Intramural Research Program | ||||||||||||||
| Direct | 127 | 3 | 130 | 130 | 3 | 133 | 136 | 3 | 139 | |||||
Total: | 127 | 3 | 130 | 130 | 3 | 133 | 136 | 3 | 139 | |||||
Total: | 238 | 3 | 241 | 247 | 3 | 250 | 255 | 3 | 258 | |||||
| Includes FTEs whose payroll obligations are supported by the NIH Common Fund. | ||||||||||||||
| FTEs supported by funds from Cooperative Research and Development Agreements. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| FISCAL YEAR | Average GS Grade | |||||||||||||
| 2021 | 12.9 | |||||||||||||
| 2022 | 12.9 | |||||||||||||
| 2023 | 13.0 | |||||||||||||
| 2024 | 13.0 | |||||||||||||
| 2025 | 13.0 | |||||||||||||
NATIONAL INSTITUTES OF HEALTH
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Detail of Positions 1
| GRADE | FY 2023 Final | FY 2024 CR | FY 2025 President's Budget |
| Total, ES Positions | 1 | 1 | 1 |
| Total, ES Salary | $186,878 | $196,801 | $200,737 |
| General Schedule | |||
GM/GS-15 | 28 | 28 | 28 |
GM/GS-14 | 34 | 36 | 37 |
GM/GS-13 | 65 | 68 | 70 |
GS-12 | 25 | 27 | 29 |
GS-11 | 13 | 13 | 13 |
GS-10 | 0 | 0 | 0 |
GS-9 | 5 | 5 | 5 |
GS-8 | 0 | 0 | 0 |
GS-7 | 2 | 2 | 2 |
GS-6 | 0 | 0 | 0 |
GS-5 | 0 | 0 | 0 |
GS-4 | 0 | 0 | 0 |
GS-3 | 0 | 0 | 0 |
GS-2 | 0 | 0 | 0 |
GS-1 | 0 | 0 | 0 |
| Subtotal | 172 | 179 | 184 |
| Commissioned Corps (42 U.S.C. 207) | |||
Assistant Surgeon General | 0 | 0 | 0 |
Director Grade | 0 | 0 | 0 |
Senior Grade | 2 | 2 | 2 |
Full Grade | 0 | 0 | 0 |
Senior Assistant Grade | 1 | 1 | 1 |
Assistant Grade | 0 | 0 | 0 |
Junior Assistant | 0 | 0 | 0 |
| Subtotal | 3 | 3 | 3 |
| Ungraded | 85 | 87 | 90 |
| Total permanent positions | 175 | 182 | 187 |
| Total positions, end of year | 261 | 270 | 278 |
| Total full-time equivalent (FTE) employment, end of year | 241 | 250 | 258 |
| Average ES salary | $186,878 | $196,801 | $200,737 |
| Average GM/GS grade | 13.0 | 13.0 | 13.0 |
| Average GM/GS salary | $135,297 | $142,464 | $145,258 |
1 Includes FTEs whose payroll obligations are supported by the NIH Common Fund.

